application
Thyroid Nodules
Americans have some form of thyroid disease, with the vast majority identified as benign lesions.1
Thyroid operations are performed each year in the United States.2
Thyroid resections are performed for benign diseases where patients could benefit from a less invasive option.3
Americans have some form of thyroid disease, with the vast majority identified as benign lesions.1
Thyroid operations are performed each year in the United States.2
Thyroid resections are performed for benign diseases where patients could benefit from a less invasive option.3
STARMED pioneered the world’s first thyroid-dedicated RF electrode in 2004, and today our devices remain the most clinically validated thyroid RFA systems available.
As of January 1, 2025, the Centers for Medicare & Medicaid Services (CMS) established official CPT® codes 60660 and 60661 for thyroid RFA. Physicians can now confidently adopt RFA knowing that the procedure is recognized, billable, and supported by payers across the United States.
With more than 200 published studies worldwide, STARMED RFA consistently shows:
A multi-institutional cohort study (2019–2022) demonstrated median volume reduction rates (VRR) of 73% at 6 months and 76% at 12 months following thyroid RFA – evidence of consistent, long-term efficacy across diverse clinical settings.
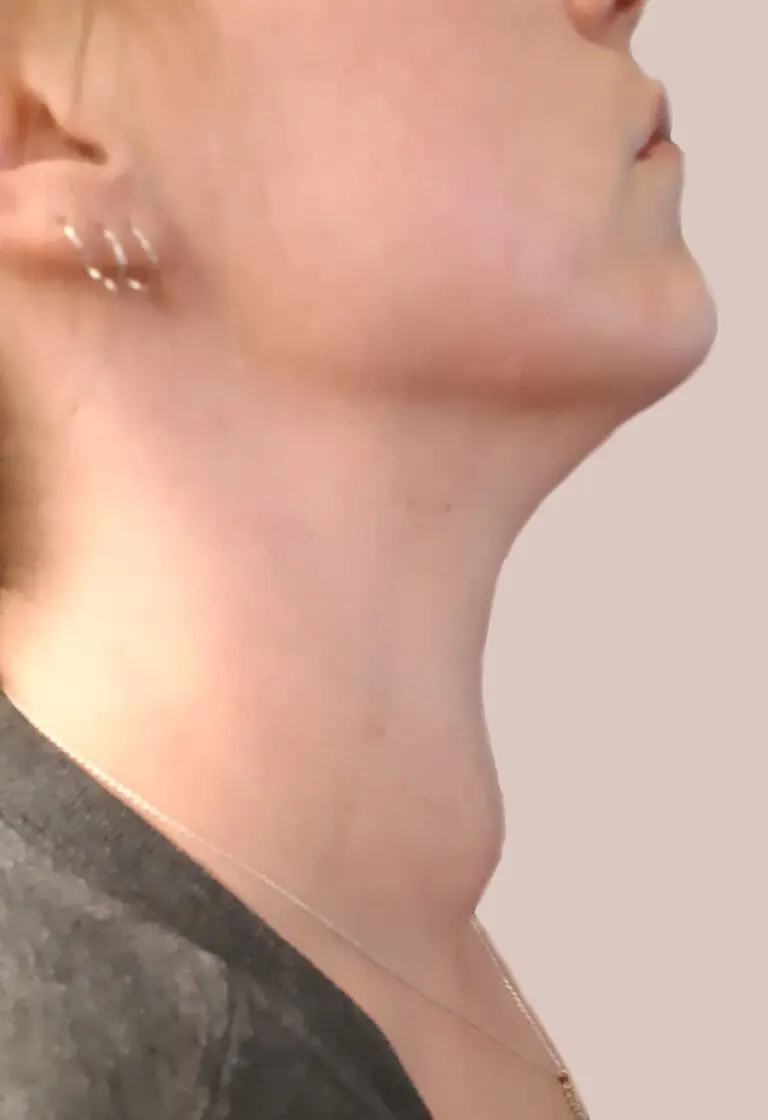



Individual results may vary. Photo courtesy of Dr. Jung Hwan Baek
Featured physician Story
Hear from Dr. Gregory Randolph, Dr. Daniel Russell, and Dr. William Kelly at Massachusetts Eye and Ear (MEE) as they share their experience adopting thyroid RFA into practice.
“RFA avoids general anesthesia, leaves no scar, and is performed on an outpatient basis with minimal pain. Patients often tolerate it even better than a diagnostic needle biopsy, making outcomes safe, predictable, and highly satisfying for both patients and physicians.”
When evaluating treatment options, physicians must balance efficacy, safety, thyroid preservation, and patient quality of life. The table below highlights they key clinical differences between radiofrequency ablation(RFA) and surgery for benign nodules.
| Clinical Procedure | RFA | Surgery |
|---|---|---|
| Settings & Anesthesia | Outpatient, ambulatory procedure Local anesthesia | Inpatient, only done in the Operating Room(OR) General anesthesia |
| Procedure Time | < 1 hour per case | Multiple hours per case |
| Recovery7 | Return to daily activity in days | 2-3 weeks recovery |
| Efficacy | 50-90% VRR at 12 Months | Complete excision |
| Thyroid Function8, 9 | Preserved; very low rate of hypothyroidism(~0%) | ~25% hypothyroidism post lobectomy, risk of hypoparathyroidism |
| Complication8, 9 | Rare: ~1.4% transient voice change(0.1% permanent) | 1-2% Recurrent Laryngeal Nerve(RLN) injury; 1-2% parathyroid injury; anesthesia risks |
| Cosmesis | Effectively scarless | Visible neck scar(unless remote access approaches) |
| Clinical Role | Minimally invasive, thyroid preserving | Standard for malignancy; invasive for benign nodules |
| Cost10, 11 | $; 30-50% of lobectomy | $$$ |
RFA provides a minimally invasive, thyroid-preserving alternative to surgery, offering comparable efficacy with fewer complications, faster recovery, and improved patient satisfaction.

Delivers constant RF output with real-time impedance feedback, ensuring safety and preventing over-ablation.

The first thyroid-specific electrode introduced to the U.S., with shorter and thinner shafts designed for safety and precision.
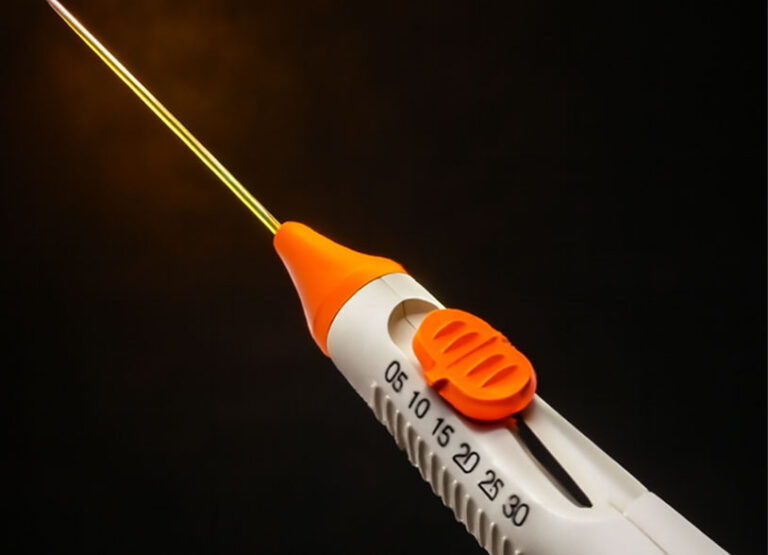
5 active tip sizes in one device, enabling tailored treatment for nodules of varying sizes.

Delivers constant RF output with real-time impedance feedback, ensuring safety and preventing over-ablation.

The first thyroid-specific electrode introduced to the U.S., with shorter and thinner shafts designed for safety and precision.

5 active tip sizes in one device, enabling tailored treatment for nodules of varying sizes.
Our U.S. Thyroid RFA Training Program offers a comprehensive learning pathway, from hands-on phantom training to live case observation and real-time proctoring. Designed and led by leading U.S. thyroid specialists, the program ensures physicians gain practical expertise, clinical confidence, and seamless integration of RFA into their practice.

Are you interested in implementing thyroid RFA treatment into your clinical practice? Reach out with your inquiry below. Our representatives will respond to your online inquiry as soon as possible.
See how safe and effective STARMED’s industry-leading equipment is in practice.

By submitting this form, you consent to receive marketing emails from STARMED America at the email provided. You can unsubscribe at any time. Please see our privacy policy for more information.